Key Takeaways:
- Orphan drug credits offer a significant tax incentive to encourage pharmaceutical companies to develop treatments for rare diseases.
- The tax credit is worth up to 25% of qualified clinical testing expenses.
- It is possible for companies to claim both R&D and orphan drug credits in the same tax year, maximizing support for a broad range of medical research.
—
Developing new pharmaceuticals and bringing them to market is an expensive endeavor. Uncommon diseases and conditions, often called “orphan diseases,” affect small populations in the United States. Given the high costs of research and development (R&D), this limited patient base can make treatment development less economically attractive for pharmaceutical companies.
Congress passed the Orphan Drug Credit (IRC Section 45C) to address this challenge and encourage the development of treatments for less profitable drug therapies.
What Is the Orphan Drug Tax Credit?
The Orphan Drug Credit is a federal tax credit designed to encourage pharmaceutical companies to invest in the research and development of treatments, cures, and preventive measures for rare diseases or conditions.
Before the Orphan Drug Act was enacted in 1983, life sciences companies often hesitated to invest in development costs for rare diseases because the small populations of potential patients made it difficult to recover development costs.
If your testing qualifies, the nonrefundable tax credit equals 25% of qualified clinical testing expenses (QCTEs) for the current taxable year.
In general, you can claim the credit between the date the U.S. Food and Drug Administration (FDA) grants the orphan drug designation and the date it approves the drug for patients.
Orphan Drug Tax Credit Eligibility Criteria
To qualify for the Orphan Drug Credit, you must first receive an orphan drug designation from the FDA. This designation is granted to drugs intended for the treatment, diagnosis, or prevention of diseases or conditions that affect fewer than 200,000 people in the United States or that affect more than 200,000 people but are not expected to recover the costs of developing and marketing a treatment drug.
Once receiving the orphan designation, you can deduct 25% of qualified clinical testing expenses incurred in the U.S.
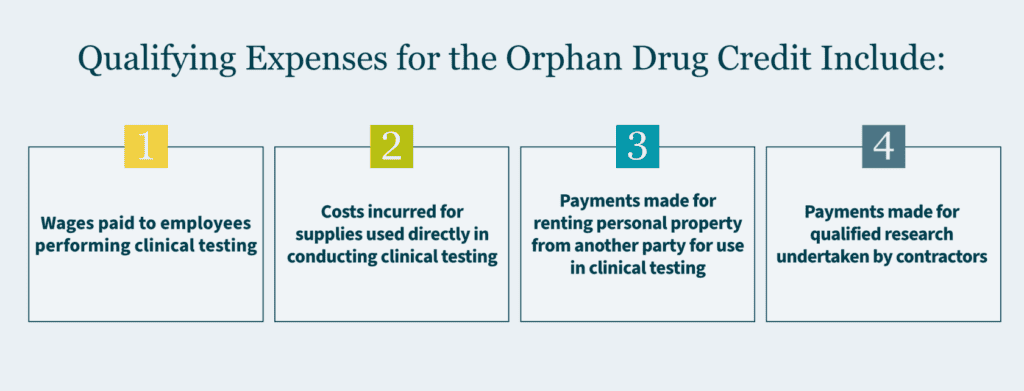
Eligible expenses are similar to those that qualify as research and experimental expenditures for the R&D credit. Some examples include:
- Wages paid to employees performing clinical testing
- Costs incurred for supplies used directly in conducting clinical testing
- Payments made to another party for computer hosting and leasing pertaining to clinical testing activities
- Payments made for qualified research undertaken by contractors
Research activities funded by a government entity or another entity other than the taxpayer do not qualify for the tax credit.
Orphan Drug Tax Credits Versus R&D Tax Credits
The Orphan Drug Credit and the R&D credit (IRC Section 41) offer substantial financial incentives to spur U.S.-based research. However, the Orphan Drug Credit provides a greater incentive than the R&D credit.
If you are researching non-orphan diseases, you can claim the standard R&D credit, which on average results in an overall credit benefit of roughly 10% of the qualified expenses.
Other benefits of the Orphan Drug Credit include claiming 100% of qualified contract research expenses, compared to 65% under the R&D credit, and claiming the credit on foreign clinical testing expenses provided the testing meets certain criteria. The criteria include:
- The testing is conducted outside the United States because there is an insufficient testing population within the U.S., and
- The testing is conducted by a person inside the United States or any other person not related to the taxpayer to whom the designation under Section 529 of the Food, Drug and Cosmetic Act applies.
Ideally, you can claim the Orphan Drug credit on qualified clinical testing expenses until the FDA approves your drug for patients, then claim the R&D credit for any ongoing qualified research expenses post-FDA approval.

Is it Possible to Claim Both the R&D Credit and the Orphan Drug Tax Credit in the Same Tax Year?
Yes, you can claim both credits in the same tax year, provided you meet the eligibility criteria for each. However, the expenses used to claim the Orphan Drug Credit cannot also be used to claim the R&D credit. The return on investment is greater under the ODC, so a company with an eligible orphan drug designation would likely pursue the ODC credit pertaining to those expenses. A company may have expenses pertaining to a non-ODC program, or expenses incurred prior and post the ODC eligible timelines, that would benefit under the R&D credit.
How MGO Can Help
The Orphan Drug Credit provides significant incentives for pharmaceutical companies developing treatments for rare diseases, making these endeavors more financially viable.
As with any tax credit, the qualifications for claiming the Orphan Drug Credit are nuanced, and it’s critical to have the necessary documentation to calculate and substantiate your claim.
If you need help determining which expenses qualify, calculating your credit, or determining how it works in tandem with the R&D credit, call or contact MGO online today. We’re happy to help you identify potential qualified expenses and maximize the tax benefits of bringing these new drugs to market to treat rare diseases.